I’m interested in the idea of replacing animal models with human-derived ways to assess drugs. Animal models make sense in that we share evolutionary history, but that’s not the same as human testing.
One new approach is to use microphysiological systems (MPS), colloquially known as “organ-on-a-chip” (OOC).
Why MPS
To back up for a moment, identifying and quantifying a drug candidate goes something like this:
- Test a molecule against a cell (in vitro, “in glass”).
- Move on to the animal models (in vivo, “in life”) for safety and efficacy.
- Potentially work up through mice, then rabbits, and so on, before first in human testing.
Wouldn’t it be better if you can skip all that animal testing? Faster, cheaper, and more accurate if it’s based on human cells and organs. That’s where organs-on-a-chip come in.
What are microphysiological systems
To try to get my head around this, I attended a session on liver-on-a-chip systems presented by Dr Emily Richardson of CN Bio and Dr Rhiannon Hardwick of Bristol Myers Squibb.
Physically, MPS are made up of plates, potentially with built-in sensors, containing living cells, fluids, and channels. It’s a dynamic environment, attempting to mimic an organ.
I get the sense this set up is bespoke at the moment for what you want to test, but there’s likely to be off-the-shelf “liver” or “lung” you can buy, if not already.
You pop them into what looks like an oven, and they do their thing, pumping fluids around and whatnot.

How they are used (or not used)
It seems to go like this: you acquire your human cells, set up the plates, drop in your drug or whatever it is you want to test. It sounds much harder than that, but it seems to be the basic idea.
After some period of time, you measure what you want to measure. And there you go: you’ve tested your drug on a human-like system.
Sounds good. I asked what was holding back these systems from becoming the preferred method for drug development. It boils down to confidence.
In particular, there’s data on compounds that were toxic to animals, but we don’t know how they would have performed in humans. This creates a gap in data for assessing these technologies. Note there are examples going the other way: not toxic in animals, but toxic in humans.
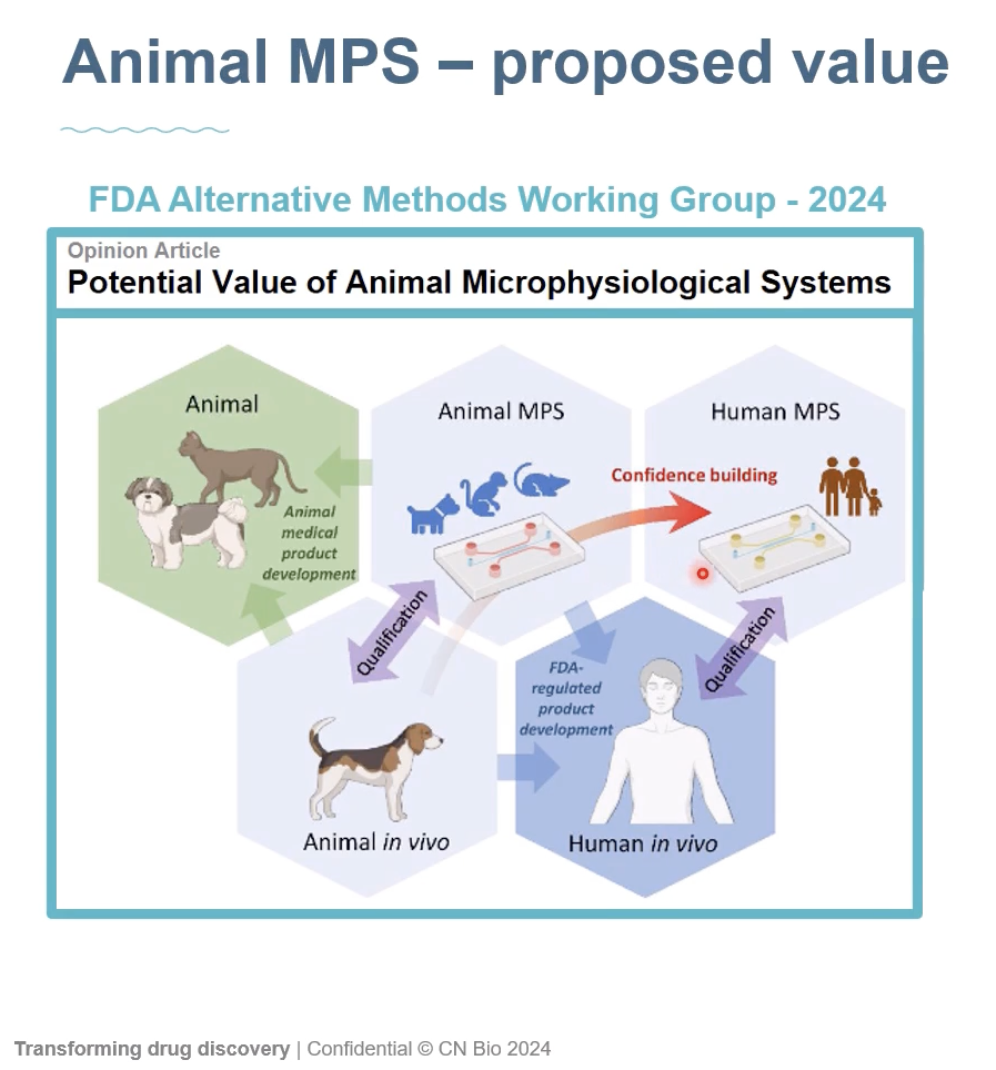
Calibrating with animal MPS
One way across this gap is by calibrating against animal MPSs. The idea is that if you have an animal MPS that accurately captures animal response, that gives you confidence that the platform (concept, system) as a whole works.
Early days, then
Such a smart idea. It reduces ethical concerns around animals, is closer to the truth by using human cells and systems, and so probably speeds up drug development.
It sounds like there’s a very long way to go on many fronts, but it’s encouraging that the FDA supports these kinds of technologies in place of animal testing.

