Targeting “senescent cells”
Reading: Senolytics target cellular senescence — but can they slow aging?, Nature Medicine, 2 Sept 2024.
Recall that senescent cells are “zombie” cells, and there’s interest in killing them off properly:
[…] senescence-targeting therapies might prove a promising avenue for the treatment of age-related diseases in the future. With several clinical trials of senolytics recruiting participants or already in progress, the next few years should provide more insight into the efficacy, safety and potential applications of such drugs.
Sounds good, but the picture may be more complex. I’m reminded of two counter views on the value of senolytics in healthy ageing, the essence of which is: senescent cells play an important role, and killing them off is maybe not so straightforward.
Earlier this year, João Pedro de Magalhãe wrote in Science:
Increasing evidence shows that senescent cells also have essential physiological functions, such as in tumor suppression, development, wound healing, tissue remodeling, regeneration, and vasculature. This raises important questions about the similarities and differences between senescent cell types and how they function in homeostasis and pathology, and it creates additional challenges in targeting them therapeutically.
In other words, how do you know if you’re hitting “good” or “bad” senescent cells?
And in 2022, David Gems and Carina C. Kern asked Is “cellular senescence” a misnomer? in GeroScience. They noted that these cells are adaptive:
It supports tissue remodeling functions in a variety of contexts, including […] acute inflammatory processes that restore normal tissue architecture and function, such as wound healing, tissue repair after infection, and amphibian limb regeneration.
They prefer the phrase “remodelling activation” to “cellular senescence”, and (as I read it) suggest that removing these “zombie cells” is missing the point. What is driving inappropriate cellular remodelling in the first place?
Figure 1 from that paper summarised this alternative. The current thinking at the top. Below is the new thinking (B), that inappropriate activation of remodelling is the issue rather than senescent cells themselves.
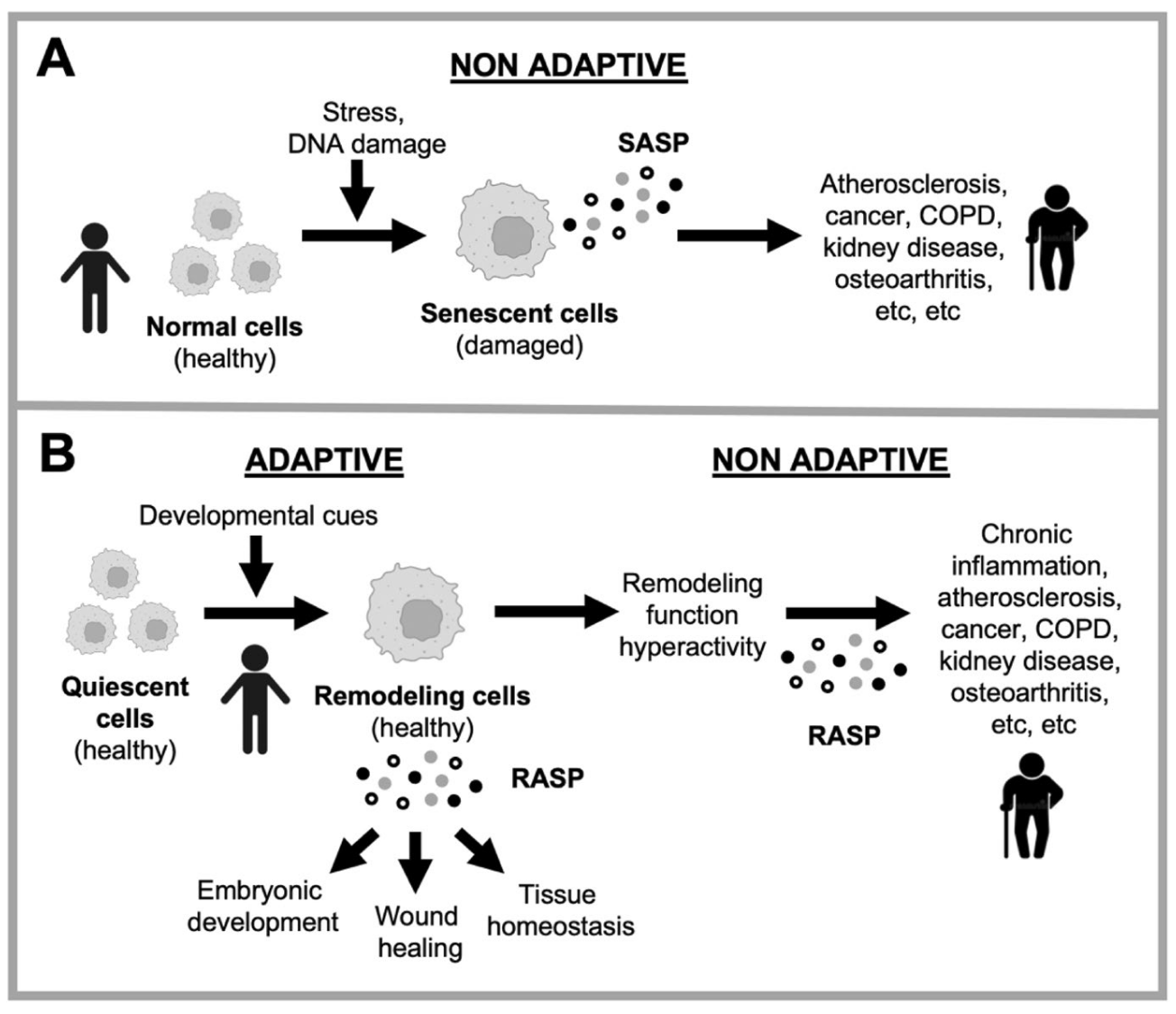
But: “the relative importance of damage accumulation vs programmatic mechanisms in late-life increases in senescent cells remains an open question.”
We’ll see how these two views play out. Either it’s “clear out the damaged cells!" or it’s figuring out why cell remodelling goes awry with age. The clinical trials will help figure this out.
